
Insulet has announced the FDA approval of their new Omnipod® 5 insulin delivery system for use in people with type 1 diabetes who are two-years-old and older. With this device, Insulet will join Tandem and Medtronic in offering an Automated Insulin Delivery (AID) system.
The Omnipod® 5 is the first insulin delivery system to require
- No multiple daily injections
- No tubing
- No finger pricks*
- No PDM.
*Finger pricks should always be used if your symptoms don’t match the reading on your G6.
What is an AID system?
In the case of Insulet, their Automated Insulin Deliver system means integrating the Dexcom G6 Continuous Glucose Monitoring (CGM) System and a compatible smartphone (or your PDM) to automatically adjust insulin. This helps to protect against highs and lows.
Who can use the Omnipod® 5?
The Omnipod® 5 is approved for use in the United States for individuals six years old and older who have type 1 diabetes.
What is new in the Omnipod® 5?
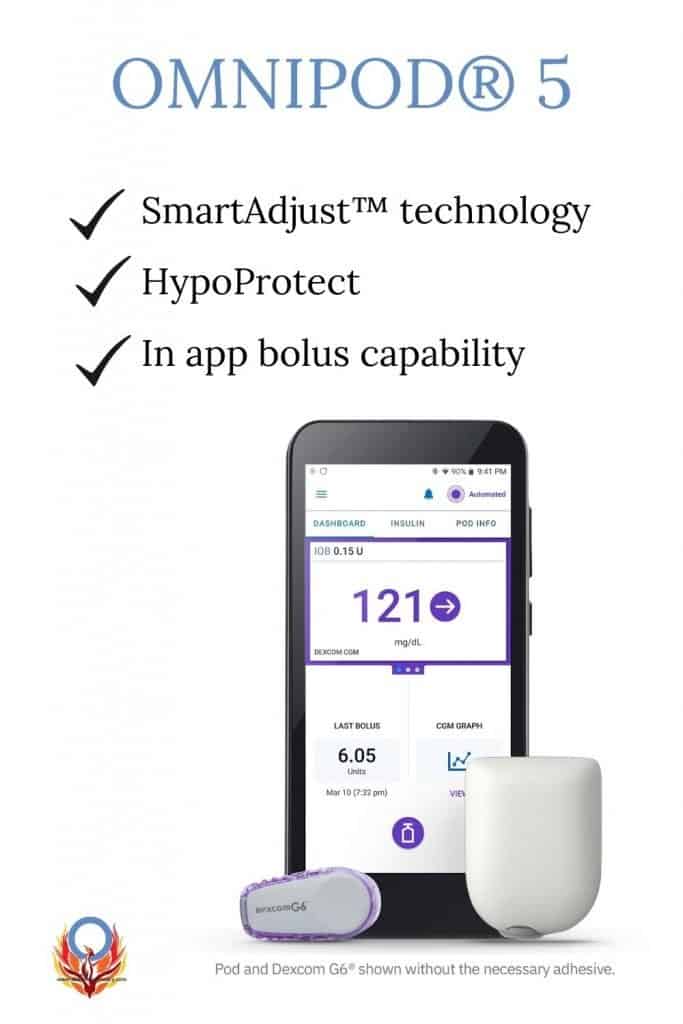
The Omnipod® 5 remains a tubeless, pod system. The Bluetooth™ pods that were released with the DASH, now have been enhanced with SmartAdjust™ technology! This means that the pods can now work with the Dexcom G6 CGM, and the Omnipod® 5 mobile app (no PDM controller required)!
SmartAdjust™ receives a Dexcom CGM value and trend every five minutes. It then predicts where glucose will be 60 minutes into the future. Finally, the system increases, decreases, or pauses insulin delivery using the user’s customized glucose target, helping to protect against highs and lows. The target range can be different depending on time of day.
DiabetesMine noted in an earlier article that the Omnipod® 5 will also include a “HypoProtect” feature. This feature is geared to prevent lows during exercise by temporarily setting the target to 150 mg/dL and minimizing basal insulin delivery.
Over the course of two or three Pods, the algorithm will begin to make more aggressive self-adjustments based on the results of the past several days. It will have a new foundational basal program set after about nine days.
Can you use the Omnipod® 5 without a CGM?
Yes, according to diaTribe, the Omnipod® 5 has three options for use.
You can use it in “automated mode”. This means that it will adjust insulin delivery automatically with a CGM connection.
You can use it in “manual mode”. In this case, the system will deliver insulin at pre-programmed rates regardless of whether the CGM is connected or not.
Finally, there is “activity mode”. This is HypoProtect feature previously mentioned. It allows the user to temporarily set a glucose target of 150 mg/dL and suspend insulin delivery during exercise or other periods of increased insulin sensitivity or high risk of hypoglycemia.
Which cellphones can be used with the Omnipod 5?
At the moment, the Omnipod 5 website is showing that the remote bolus option is only available for the Samsung Gallaxy S10 (4G). You can check this link to see when more options are available.
What stays the same?
While the Omnipod® 5 comes with some incredible new features, it has kept many features that Podders have come to love.
It remains the same shape and size as the current pods. It can be worn for up to 3 days and holds up to 200 units of insulin.
If you don’t want to use a cellphone, you will be able to access a free Omnipod® 5 controller with your first prescription.
What else can we expect?
There is more coming for the Omnipod® 5! Insulet has promised that the Omnipod® 5 will work with the Freestyle Libre at one point in the future.
Can I upgrade from the Omnipod DASH® ?
Yes! According to the Insulet press release, if you are using the Omnipod DASH® , you will be able to upgrade to the Omnipod® 5 at no additional cost.
When can I get my Omnipod® 5?
According to Insulet, “Omnipod 5 will launch through the pharmacy channel, providing customer benefits of no contract, no commitment, and no obligation. In the coming days, Insulet will offer Omnipod 5 in a limited market release so that the Company can incorporate learnings to deliver a best-in-class product experience. Omnipod 5 is expected to be broadly available shortly after the limited market release.”



I am very interested in getting this new Omni Pod as I have just started on the Dexcom6
P,ease let me know where I can order it.
Please contact your local OmniPod rep if living in the US
Confused by The Omnipod® 5 is the first insulin delivery system to require
No multiple daily injections
No finger pricks*
No insulin pump requires multiple daily injections and tandem does not require finger pricks either.
Excellent.
Is it available in Canada?
If yes, where?
if not, when will it be available
I’m Dexcom G6 user.
Sadly it isn’t approved yet but hopefully soon! We will let you know.
When will it be available for type 2 diabetics in the US
We will let you know as soon as we hear.
I just need more insulin than 200U. When do I need that? Rarely. But rarely isn’t never. A few weeks ago, I went to 210U in three days. It has always freaked me out that I might run out of insulin. The second issue is the controller. I am convinced I would lose it or forget it, and then I would be up the creek without a pump. So to speak. 🙂
Will the Pod work with all fast acting insulin? Apidra, Fiasp, Humalog, Lyumjev ect?
All insulin pumps only use rapid acting insulin like Humalog or NovoRapid (Novolog). Not all rapids are approved for use but you can speak with your diabetes team to see if the one you are using has been approved for use in the Omnipod.
We hope that it is soon compatible with the iPhone .
I agree!!! Fingers crossed!
I agree as well!
Just seen commercial. Is it compatible with the iPhone yet?
Does the Omnipod 5 communicate with the Libra 2?
It doesn’t yet. That is something that is being worked on between the companies.
Why is it so unclear as to whether or not the Omnipod (any version) is covered by insurance? Every other health product has that answer on the first day. Yet, Omnipod appears to favor using its “pharmacy outlets” to sell its products for $500/month. I dont need it at that price. My a1c has been at 7 for many, many years. Id like to try omnipod, but not at $500 / month.
Insurance seems to vary country to country, provider to provider. It is tragic that cost and insurance become a barrier to access for so many.
I am looking for a discussion group among people who are using the Omnipod 5. So far, I am very disappointed in the Automated operation. I have discussed with clinical support at Omnipod and with my doctor (who has trouble getting trained by Omnipod) without enough helpful information. Please let me know if there is such a discussion group available.
Thanks.
I am sorry to hear that you aren’t getting the support that you need from Omnipod. Have you considered Omnipod groups on Facebook or Reddit? They are often filled with users who have had similar experiences. Diabetes Advocacy will be opening a VIP area in the coming months that will offer a place to share information as well.
Y is Insulet so concerned with connecting to Libra and not correcting current issues?????
1. Get rid of extra screen to enter bg, all information should be on one screen carbs, bg and calculation. Start up is tooooo slow when calling tec support staff is impatient, but to bad the controller is slow!!!!
Why can’t the iPhone be used as a controller yet?????? Oh because they r to busy trying to connect Libra cgm!!!
Insulets statement “working to simplify life for people with diabetes”. Bogus. The Omnipod 5 operation was slightly flawed, now after the update 1 it sucks! To many screen to enter information when it coul be on one screen.
Due to all the different screens insulets impatient staff treats patients like idiots!!!! Some don’t comprehend patients have used Omnipod for many years n know more than they do!!!
Over the years I have talk to trainers that had no clue what I was talking about because their training controller didn’t work like the patients controller!!!!!
Insulet needs a competitive company in the US to make them work harder for a better product.
Omnipod doesn’t hold to their moto, simplify life for people with diabetes……bogus!
Every update on the controller for Omnipod 5 has caused more button pushing to do a simple bg reading entry!!!
Omnipod says no PDM, again bogus…..not all phones can be used so you need a PDM!
Omnipod claim to listen to it’s customers about issue, if the listened they would have fixed the update issues and the Dexcom G7 would have a lot better communication with the pod since it’s not possible to keep both devices (pod and sensor) on the same side all the time!!! Pod changes are every 2-3 days depending on insulin needs and sensor is every 10 days! Look up the so called placement locations of each……we have to keep rotating placements to keep from having other issues!
The only way to resolve this issue is a 10 day pod which is unrealistic due to other issues such as infections which already happens for some people!!!!!!
Insulet needs to stop all the lies about the Omnipod product!!!!!
Please consider making a larger version of pods. This 200 unit capability is frustrating. Insulin usage varies in every person for different reasons. I have to change my pods every 48 hours and still need to bolus via pen (which defeats the IOB tracking) 300 unit capability would be so much better for alot of patients. Otherwise this system is almost perfect.
Does the omnipod 5 work with older pods?
No it does not.